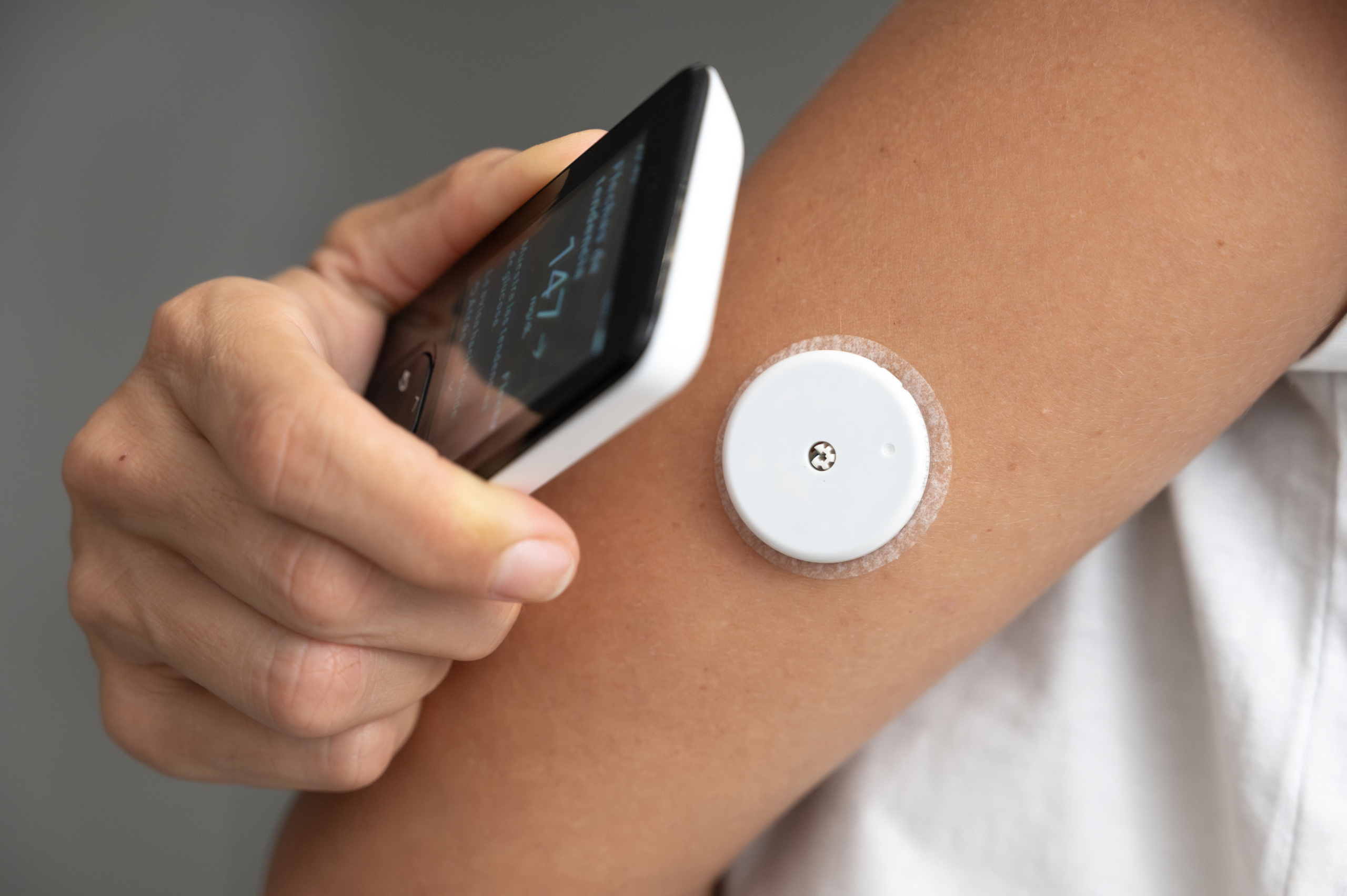
Wearable medical devices and remote patient monitoring systems are transforming healthcare by enabling continuous, real-time health tracking outside traditional clinical settings. At the heart of these life-saving technologies lies sophisticated printed circuit board assembly (PCBA) design and manufacturing. As a global leader in technical manufacturing, E-BI produces high-reliability medical-grade PCBAs in its facilities across China, Vietnam, and Thailand. This article explores the critical role of advanced PCBA design in wearable medical devices and how E-BI’s expertise powers this rapidly growing sector.
The Rise of Wearable Medical Devices
The global wearable medical device market is expected to reach USD 174.5 billion by 2030, growing at a CAGR of 26.1% from 2023 Grand View Research. Devices such as continuous glucose monitors, ECG patches, smartwatches with SpO₂ and heart-rate monitoring, and remote vital-sign trackers rely on ultra-compact, low-power, and highly reliable electronics to deliver accurate data 24/7.
Key PCBA Requirements for Wearables
Wearable medical PCBAs must meet stringent requirements:
- Miniaturization – often smaller than 20×20 mm
- Ultra-low power consumption for multi-day battery life
- High-density interconnect (HDI) and flexible/rigid-flex designs
- Biocompatibility and skin-safe materials
- ISO 13485 and FDA-compliant manufacturing processes
Advanced techniques like 01005 components, embedded passives, and chip-on-board (COB) assembly are now standard Jabil.
Remote Monitoring and Wireless Connectivity
Modern wearables transmit data via Bluetooth Low Energy (BLE), Wi-Fi, or cellular networks to cloud platforms and clinician dashboards. Some emerging applications are adopting LoRa (Long Range) technology for low-power, long-distance transmission in rural or campus-wide hospital settings, enabling seamless monitoring over several kilometers without relying on cellular infrastructure Semtech – LoRa. This is particularly valuable for post-discharge patients or elderly care facilities.
E-BI’s Medical-Grade PCBA Capabilities
E-BI operates ISO 13485-certified facilities in China, Vietnam, and Thailand, specializing in high-reliability, high-mix/low-to-medium volume medical electronics.
Miniaturization and HDI Expertise
Our facilities deploy 4–16 layer HDI boards with micro-vias, via-in-pad, and stacked via technology to achieve the smallest possible footprints. We routinely place 01005 components and use rigid-flex construction for devices that must conform to the body Assembly Magazine.
Low-Power and Wireless Integration
E-BI integrates BLE, NFC, and LoRa modules directly onto medical PCBAs, optimizing power management ICs and antenna layouts for maximum range and battery life. Our cleanroom environments (Class 100,000 / ISO 8) ensure contamination-free assembly of skin-contact devices.
Regulatory Compliance and Traceability
Full IPC-6012DS Class 3 and IPC-A-610 Class 3 standards are followed. Every component is lot-traceable, and we provide complete Device History Records (DHR) required for FDA submissions U.S. FDA.
Challenges in Wearable Medical PCBA Design
Key challenges include:
- Achieving weeks of battery life while maintaining continuous monitoring
- Ensuring biocompatibility and sweat/skin resistance
- Managing heat in skin-contact devices
- Meeting stringent EMC/EMI requirements in a tiny form factor
E-BI addresses these through advanced thermal simulation, conformal coating, and shielded flexible circuits Wiley.
Powering the Future of Healthcare with E-BI
Advanced PCBA design is the foundation of reliable, life-changing wearable medical devices and remote monitoring systems. E-BI’s ISO 13485-certified manufacturing in China, Vietnam, and Thailand delivers the precision, miniaturization, and regulatory compliance that medical OEMs demand.
For companies developing next-generation wearables—whether using BLE, cellular, or emerging LoRa-based solutions—partnering with E-BI provides a strategic advantage. Our proven medical electronics expertise can accelerate your time-to-market while ensuring the highest quality and patient safety. Connect with E-BI today to bring your wearable medical vision to life.